The Test Is Not the Intervention
A Clinical Design teardown of ctDNA and the treatment decision it was built to change.
Clinical Design Teardown #1
1. The decision before the test
At week five after surgery, the crucial clinical question is whether this patient should receive months of chemotherapy for a risk that may no longer be there. How is this tested? Whether tumour DNA can be found in their blood. The decision depends on this test.
However, this leads us to a key realization: precision oncology often starts with the test. Clinical Design, however, starts with the decision.
The test is not the intervention, and should not be considered so. The changed decision is.
A test reports tumour DNA in the bloodstream. A changed decision tells us who gets chemotherapy on a Thursday morning, in the right time window, with measurable consequences. This piece is about the second one.
A previous edition of The Clinical Decade introduced the operator architecture: the care-changing unit as the object, the Clinical Design Loop as the operating sequence, AEIOU as the constraint scorecard.1
This is the first Clinical Design Teardown, a series where we run the Clinical Design methodology on biotech and techbio innovations.
Today, this is one of the clearest worked examples of a biomarker becoming a pathway strategy: postoperative ctDNA, in resected stage II colon cancer, deciding whether a specific patient should receive adjuvant chemotherapy.
For postoperative ctDNA in resected stage II colon cancer, the Clinical Design read is this:
• The object: in resected stage II colon cancer, after surgery and before the adjuvant decision, the medical oncologist uses postoperative ctDNA status to recommend adjuvant chemotherapy or observation.
• The ladder: signal (Tie 2016), decision (DYNAMIC 2022), durability (DYNAMIC 5-year 2025), scale (GALAXY/CIRCULATE-Japan 2024), governance (NCCN/COBRA/DYNAMIC-III).
• The pivot: DYNAMIC did more than validate a biomarker. It changed the chemotherapy denominator.
• The discipline: prognostic ≠ predictive ≠ action-changing. The three claims should never share a slide unless distinguished.
• The operator rule: don’t pilot the test. Pilot the care-changing unit.
2. The wrong object: we keep evaluating the test
A common error in healthcare innovation reviews is to start with the technology and look for a decision to influence. That order rarely survives contact with a busy clinic.
The cleaner starting point is the decision, and what we already know about it.
In resected stage II colon cancer, the existing decision is whether to give adjuvant chemotherapy after surgery. Clinical guidelines have long framed this as anything but routine: stage II is a heterogeneous group, and adjuvant chemotherapy is recommended selectively, weighted by T stage, nodal sampling, MMR/MSI status, lymphovascular invasion, perforation, obstruction, and shared decision-making with the patient.2
Most stage II patients will be cured by surgery alone. A subset will recur. Adjuvant chemotherapy reduces recurrence on average, but the toxicity is real, and the same drug regimen produces very different value depending on a patient’s actual recurrence risk. The result is the kind of decision oncology has long called uncomfortable: oncologists, MDTs, patients and families are all forced to reason under uncertainty about a treatment that will harm everyone in the short term and help only some in the long term.
ctDNA matters because the pathway already contains a decision that is uncomfortable, variable, and consequential. The test arrives because the decision was already imperfect.
If the decision did not exist, no biomarker, however elegant, would create one. If the decision existed but no one could act on it, no biomarker, however accurate, would change care. The decision is the gravity. Everything else moves into orbit around it.
3. The care-changing unit for ctDNA
The care-changing unit has six slots: a population, a decision point in a pathway, a named actor with the authority to act, a concrete action, a clinically meaningful time window, and a measurable consequence.
The careless framing of ctDNA is “ctDNA can be measured in stage II colon cancer.” That sentence describes an analytical capability. It does not describe care.
The care-changing unit reads differently:
In resected stage II colon cancer, after curative-intent surgery and before the adjuvant therapy window closes, the medical oncologist uses postoperative ctDNA status to recommend adjuvant chemotherapy or observation, with the goal of reducing unnecessary chemotherapy while preserving recurrence-free survival.
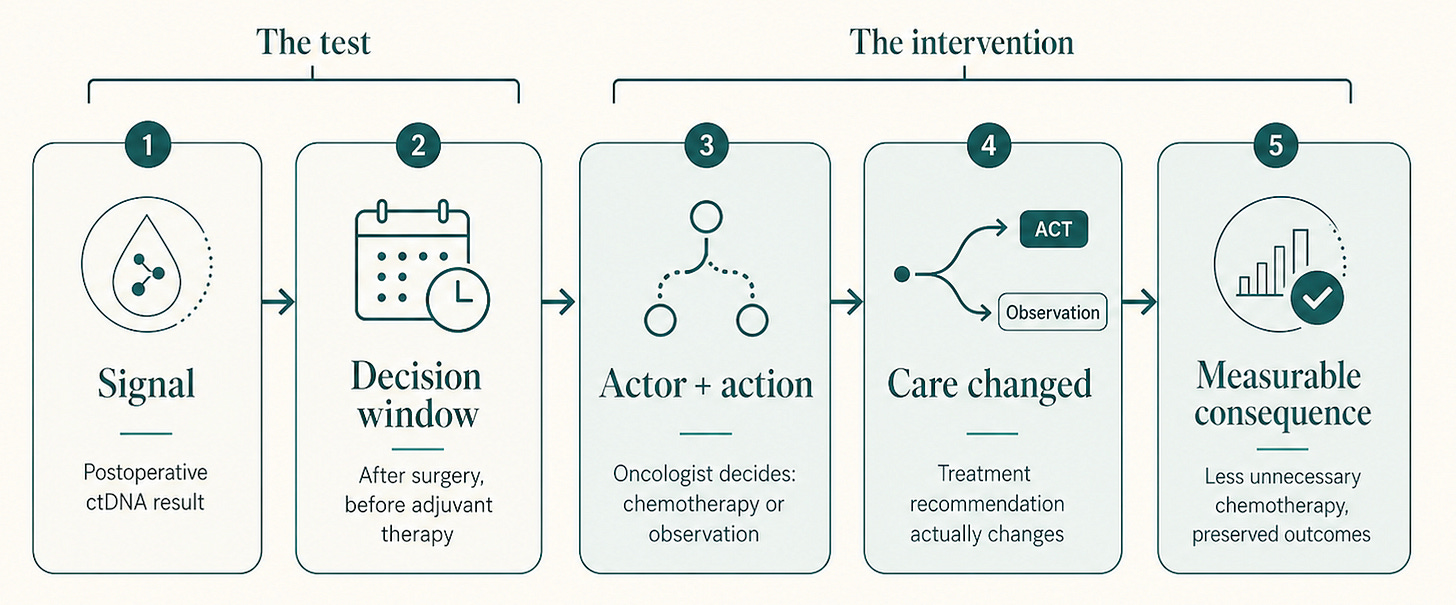
Six slots, filled.
• Population: patients with resected stage II colon cancer.
• Decision point: the post-surgery, pre-adjuvant decision.
• Actor: the medical oncologist, in some systems supported by the colorectal MDT.
• Action: recommend adjuvant chemotherapy, recommend observation, or, in some clinical scenarios, escalate to a doublet regimen.
• Window: the result has to land before the adjuvant decision. In DYNAMIC, ctDNA was measured at week 4 or 7 after surgery.3
• Consequence: less adjuvant chemotherapy used in the population, preserved recurrence-free and overall survival, lower treatment exposure and patient burden, and a credible economic case where the payment architecture allows value capture.
Notice what the sentence forces. It forces an actor, not a department. It forces a window, not “as soon as possible.” It forces a consequence, not a vague “improve outcomes.”
A ctDNA result that arrives after the adjuvant decision was made stops being precision oncology and becomes post hoc biology.
That distinction is small in print and consequential in pathway design. Most ctDNA innovation that struggles in implementation struggles here, in the clinical-moment placement: the assay is sensitive, the report is informative, and the result lands too late to matter.
Strictly, ctDNA is not a single intervention. Assay design, tissue requirements, turnaround time, report format, and failure rate all matter. In this teardown, ctDNA is shorthand for a postoperative ctDNA pathway capable of returning a usable result before the adjuvant decision.
4. The evidence ladder: signal, decision, durability, scale, governance
The evidence on ctDNA in stage II colon cancer reads as a ladder, not a lump. Treating it as a lump is how innovation reviews keep getting it wrong.
Each rung is a different kind of claim. Each one earns a different right to act. Reading them in order is what separates a Clinical Design view from a marketing view.
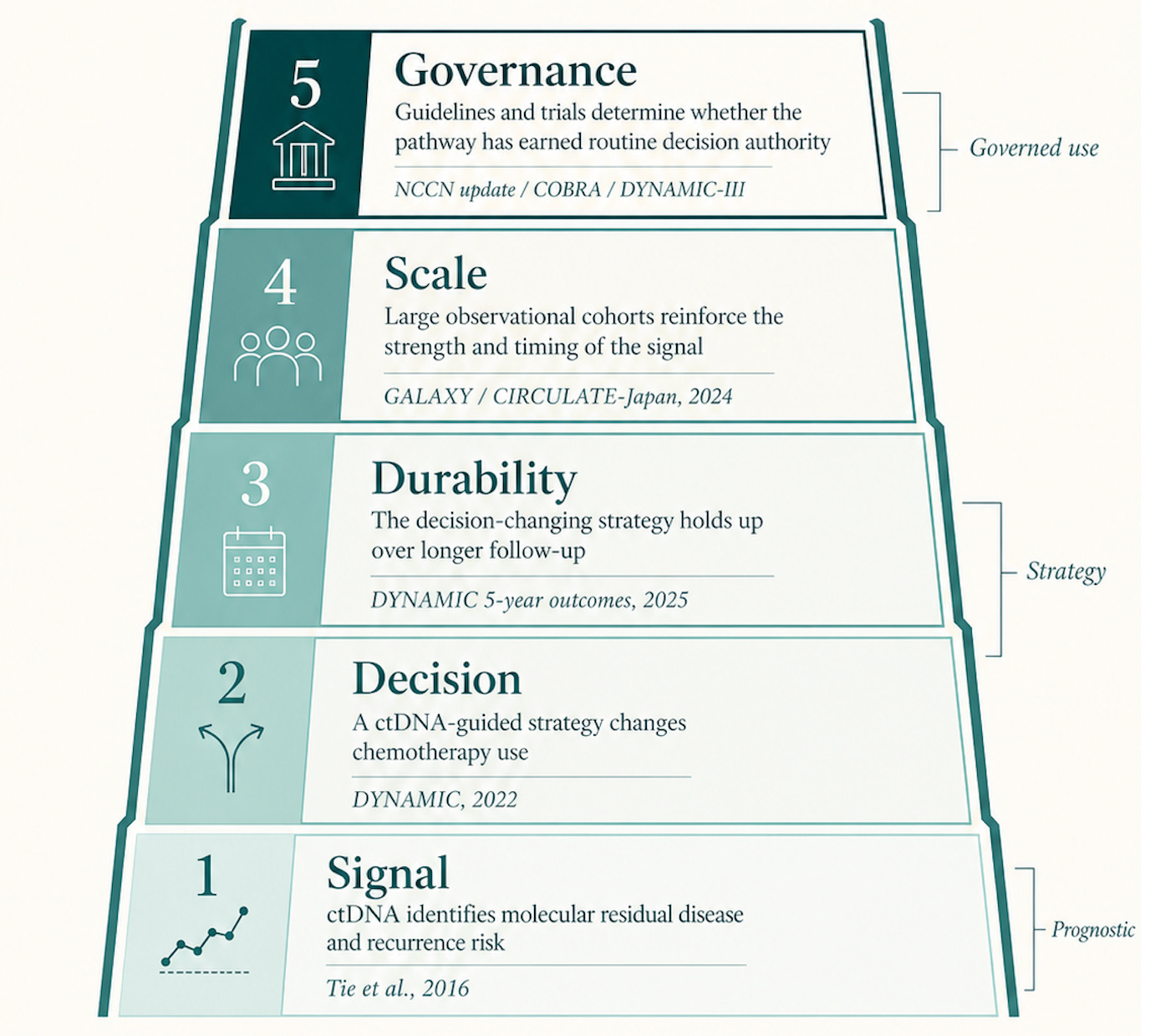
4.1 Signal: ctDNA finds residual risk
The base rung is signal. Tie et al. (Science Translational Medicine, 2016) showed that postoperative ctDNA detection in stage II colon cancer identified a population at very high risk of recurrence: in untreated patients, 79% of ctDNA-positive cases recurred, versus 9.8% of ctDNA-negative cases, with a hazard ratio of 18.4
That is a powerful signal. It is also still only a signal.
Prognostic information tells you who is at risk. It does not yet tell you what to do. A high hazard ratio is not a pathway. The most important sentence to put on a slide deck after Tie 2016 is the one most decks skip: this finding earns the right to design the next study, not the right to change practice.
4.2 Decision: DYNAMIC changes the chemotherapy denominator
The middle rung is decision. DYNAMIC (NEJM, 2022) is the heart of this article and one of the cleanest decision-changing ctDNA trials in the field.5
DYNAMIC randomised 455 patients with resected stage II colon cancer to either ctDNA-guided management or standard clinicopathological management. In the ctDNA-guided arm, a positive ctDNA result at week 4 or week 7 triggered a recommendation for adjuvant chemotherapy, while a negative result supported observation. In the standard arm, treatment was guided by conventional risk features.
The headline numbers were exactly what a Clinical Design reader hopes to see. Adjuvant chemotherapy use dropped from 28% in the standard arm to 15% in the ctDNA-guided arm. Two-year recurrence-free survival was 93.5% in the ctDNA-guided arm versus 92.4% in the standard arm. The non-inferiority margin held.
DYNAMIC is powerful because it did not merely ask whether ctDNA predicted recurrence. It asked a harder question: can a pathway safely treat fewer patients by binding a molecular signal to an adjuvant chemotherapy decision?
DYNAMIC did more than validate a biomarker. It changed the chemotherapy denominator.
That sentence is the reason this trial belongs at the top of every Clinical Design syllabus on ctDNA.
A precision note matters here. DYNAMIC is best read as pathway-strategy evidence, not strict predictive evidence. ctDNA-positive patients were not separately randomised to chemotherapy versus no chemotherapy inside the trial, so the design cannot prove that ctDNA predicts chemotherapy benefit for every treatment action. The claim it does prove is enough for Clinical Design: a ctDNA-guided strategy reduced chemotherapy use without compromising recurrence-free survival.
4.3 Durability: the five-year follow-up
The third rung is durability. Early non-inferiority is useful. Durable outcomes are what make a redesigned decision credible.
The DYNAMIC five-year results (Nature Medicine, 2025) reported, with median follow-up of 59.7 months, 5-year recurrence-free survival of 88% in the ctDNA-guided arm versus 87% in the standard arm, and overall survival of 93.8% versus 93.3%. Among ctDNA-positive patients who received adjuvant chemotherapy, 35 of 40 (87.5%) showed ctDNA clearance after treatment. 6
The decision change held over time. The reduction in adjuvant chemotherapy did not produce a quiet excess of late recurrences in the ctDNA-guided arm.
The five-year data also suggest a second design frontier. Longitudinal ctDNA clearance or persistence after adjuvant therapy may become a second care-changing unit, focused on post-treatment surveillance, intensification, or trial enrolment. It deserves its own action contract. It should not be smuggled into the first. The first decision is whether to give adjuvant chemotherapy. The second is what to do with persistent or cleared molecular risk after treatment. Two questions, two pathways, two governance regimes.
4.4 Scale: GALAXY confirms the gravity
The fourth rung is scale. GALAXY, the observational arm of CIRCULATE-Japan (Nature Medicine, 2024), studied molecular residual disease across 2,240 patients with stage II–III resectable or stage IV colorectal cancer. MRD positivity in the postoperative period was strongly associated with worse disease-free and overall survival, and the analysis defined a postoperative MRD window of 2 to 10 weeks before adjuvant chemotherapy in which the signal is most actionable.7
GALAXY is observational. It does not replace a randomised pathway trial.
What it does is confirm that the signal is not anecdotal. At population scale, the prognostic gravity of postoperative ctDNA is consistent and reproducible. Combined with DYNAMIC, the signal is real and the decision change is workable. The larger the signal, the more dangerous it becomes to leave the action undefined.
4.5 Governance: why the caution matters
This is where the story becomes more interesting than “ctDNA works.”
DYNAMIC tests a pathway strategy under defined trial conditions. Guidelines decide whether that strategy has earned routine decision authority across heterogeneous systems, clinicians, patients, laboratories, and payment models. Two legitimate questions, marking a boundary rather than a contradiction.
The 2025 NCCN update, as summarised in OncLive reporting, recognises ctDNA as carrying prognostic information in colon and rectal cancer while remaining cautious about predictive value. In the relevant adjuvant contexts, it does not recommend routine ctDNA-based de-escalation or treatment decision-making outside clinical trials.8
Guidelines that hesitate are doing governance work. They are asking whether the biomarker has become a governed decision.
The same governance posture sharpens when the evidence pushes back. DYNAMIC-III studied stage III patients and tested both ctDNA-guided de-escalation and intensification strategies. In ctDNA-negative patients, de-escalation reduced oxaliplatin use and hospitalisations but did not meet the prespecified non-inferiority margin for recurrence-free survival; in ctDNA-positive patients, intensification did not improve outcomes versus standard management.9 COBRA (NRG-GI005), in stage IIA, was halted at an early prespecified phase II futility analysis after adjuvant chemotherapy did not increase ctDNA clearance compared with no chemotherapy in patients with detectable ctDNA.10
The biomarker can be right and the action contract still wrong.
Read together, DYNAMIC-III and COBRA work as demonstrations of method, beyond their value as cautions. Each one names a different way a ctDNA-guided strategy can break: a non-inferiority margin can fail, an action lever can be the wrong one, a setting can refuse to generalise. The same Clinical Design discipline that earned DYNAMIC its place earns DYNAMIC-III and COBRA theirs.
The ladder can be summarised bluntly:
Signal earns the right to design the action. Strategy evidence earns the right to test the pathway. Governance decides whether the pathway can scale.
5. Run the Loop on ctDNA
The Clinical Design Loop has six stages. Together they work as a failure-localisation tool, not a maturity model.
What follows is one operator pass, in table form. Each row is one stage: the question and the failure mode the literature predicts.
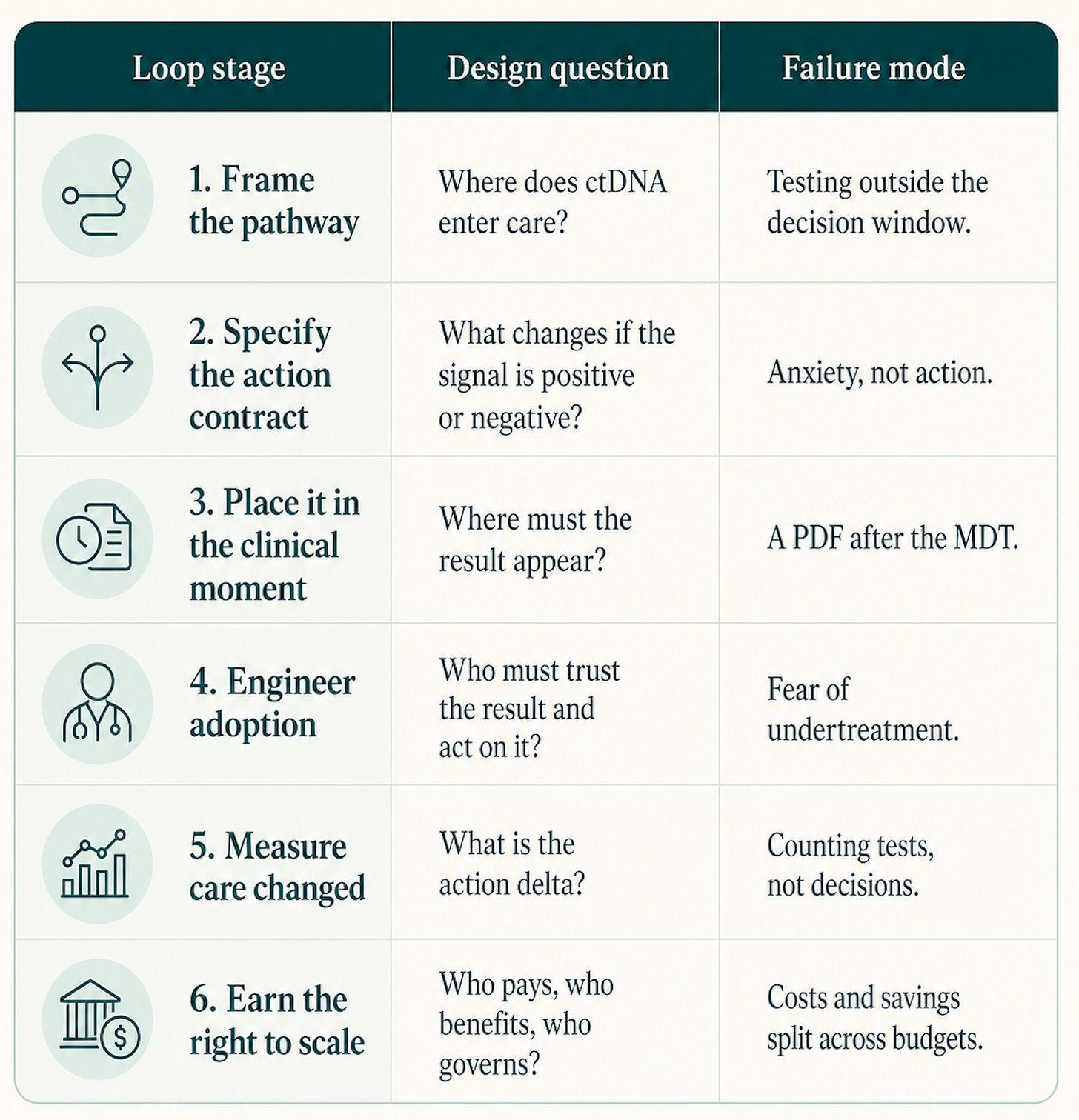
AEIOU pressure: Ownership and Unit Economics dominate stages 1 and 6; Interoperability dominates stage 3; Adoption and Evidence dominate stages 4 and 5.
A maturity model tells you how advanced ctDNA looks. The Loop tells you where it will break.
Run it before the pilot, not after.
6. AEIOU pressure on ctDNA
AEIOU is the constraint scorecard. Five vowels, scored on the case.
• Adoption: does the result change what oncologists recommend?
• Evidence: is the claim prognostic, predictive, or action-changing?
• Interoperability: does the result arrive before the decision?
• Ownership: who owns the action after the result?
• Unit Economics: who pays for the signal, and who captures the value of the changed decision?
Three vowels are tractable for ctDNA today.
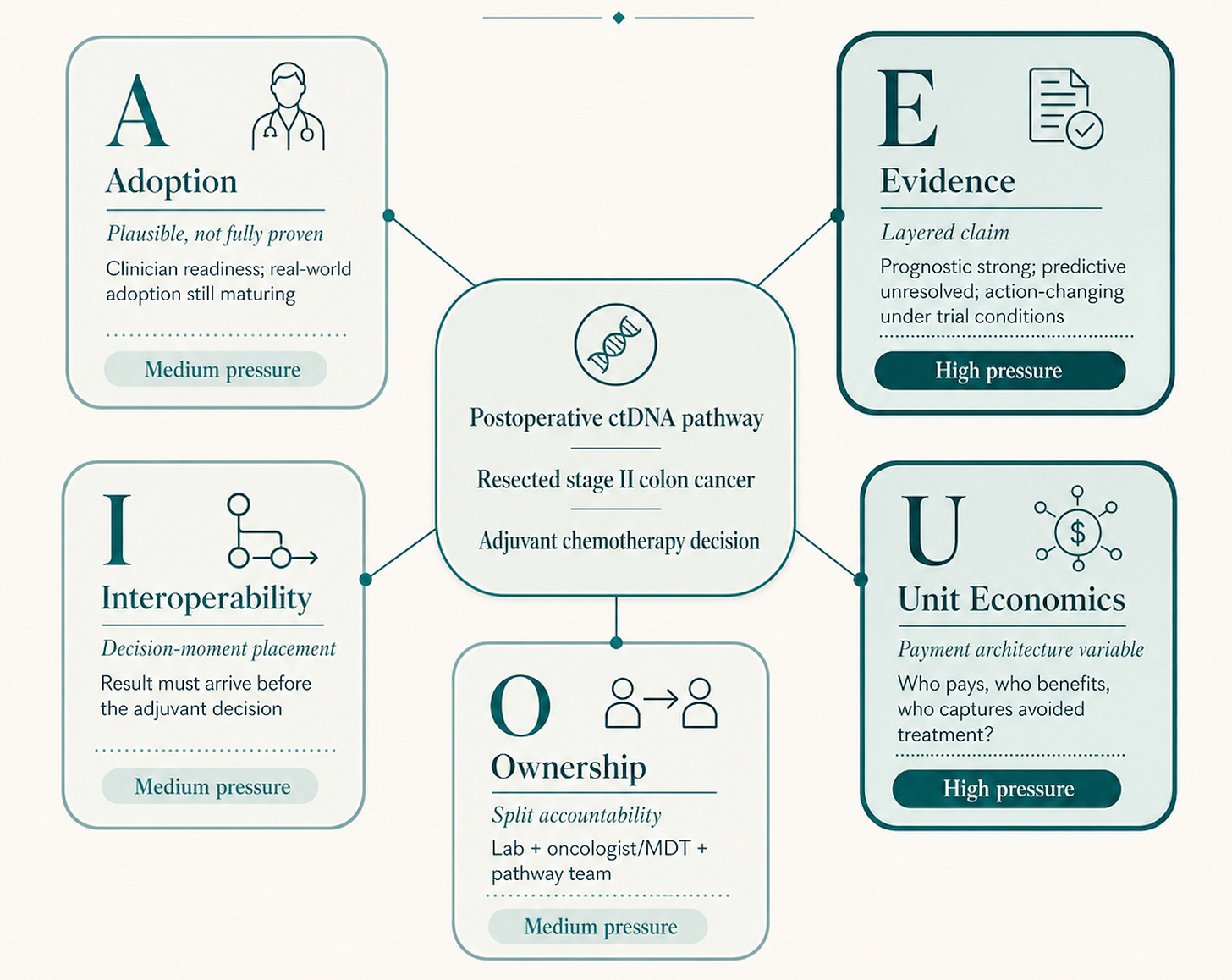
Adoption is plausible but not fully proven. DYNAMIC shows protocolised treatment separation under trial conditions, and the vignette literature suggests oncologists are willing to modify recommendations when the signal is clear.11 Real-world adoption is the next evidence claim to mature.
Interoperability is a workflow-semantic question. The result has to be placed where the decision happens, before the window closes, in a form the oncologist can use.
Ownership, in stage II colon cancer, splits cleanly between the lab (analytical validity), the medical oncologist or MDT (treatment recommendation and safety boundary), and the pathway team (order set, result-routing, audit, escalation, and what happens when the test fails to return in time).
Two vowels deserve a longer look.
Evidence: the claim hierarchy
Three different evidence claims tend to be conflated.
Prognostic evidence tells you who is at risk. ctDNA carries this in abundance: Tie 2016, GALAXY, several Japanese and US cohorts.1213
Predictive evidence tells you who benefits from the action. ctDNA does not yet carry this cleanly for the adjuvant chemotherapy decision in stage II, because no trial has randomised ctDNA-positive patients to treatment versus no treatment with sufficient power. DYNAMIC’s positive-arm randomisation was within management strategies, not within drug treatment.
Action-changing evidence tells you that, within an implemented pathway, the result changes recommendations and received care, with measurable downstream consequences. DYNAMIC supports this under trial conditions. The vignette literature suggests clinician readiness. Real-world adoption remains its own evidence claim.
Conflating these three is how slide decks accidentally promise what data does not yet support.
Unit Economics: payment architecture is the variable
A 2024 budget impact analysis from a US payer perspective estimated that a ctDNA-guided adjuvant strategy could be approximately budget-neutral at test prices around $16,202 per patient in a commercial population and about $5,793 in a Medicare Advantage population.14 A 2024 Dutch analysis found that ctDNA-augmented strategies could be more clinically effective but were not cost-effective at the chosen Dutch threshold unless the test price was lower, the test performance was higher, or the test could demonstrate predictive value.15
Two studies. Two economic verdicts. The biology has not changed between them. The payment architecture has.
The unit economics question is whether the payment architecture lets the system capture the value of the decision it changes. Test price alone does not answer it.
Avoided chemotherapy does not automatically generate a business case if the savings, incentives, and budgets sit in different places.
7. What ctDNA teaches us about Clinical Design
Run the Loop and the AEIOU scorecard, and a small set of sharp distinctions emerge. Each one is portable beyond ctDNA.
A validated test is not implemented care. Analytical validity gets the signal into the lab. Clinical Design gets the signal into a decision.
Prognostic, predictive, and action-changing are three different evidence claims. Prognostic means risk. Predictive means treatment effect. Action-changing means a clinician is willing, authorised, and measured to do something different.
Negative ctDNA still carries residual risk. De-escalation is a governed decision about acceptable risk, made transparent to the patient, with safety boundaries and follow-up. The numbers in DYNAMIC are good, not zero.
Timely is not the same as fast. A result that arrives in 7 days is fast in some pathways and useless in others. The relevant question is whether the result arrives before the clinical decision is made.
Guidelines are governance, not inertia. When guidelines hesitate, the lazy explanation is conservatism. The better explanation is that they are deciding whether a strong signal has earned routine decision authority at scale.
The biomarker can be right and the action contract still wrong. DYNAMIC-III and COBRA make this concrete: a strong signal does not guarantee that the chosen treatment lever will move the outcome.
8. Minimum Viable Care-Change Dashboard for ctDNA
Most adoption dashboards count the wrong things. They count tests ordered. They count reports issued. They count clinician logins.
A care-change dashboard counts whether the decision changed.
For ctDNA in stage II colon cancer, twelve metrics make the chain visible.
1. Eligibility denominator. How many resected stage II patients enter the post-surgery adjuvant decision window each month?
2. Test completion rate. What share of eligible patients had a postoperative ctDNA test attempted, and what share returned a usable result?
3. Failed or non-actionable test rate. What share of eligible patients never reached a usable result because of tissue unavailability, sample failure, insufficient material, logistics, cancellation, or timing? Pathway reality lives in this number.
4. Turnaround within decision window. What share of results landed before the adjuvant decision was made, within the local adjuvant decision window, with DYNAMIC’s week 4/7 testing schedule as one worked example?
5. Result reviewed at the decision moment. What share of results were seen by the medical oncologist or the MDT before the adjuvant recommendation was finalised?
6. Signal-to-action conversion. What share of positive results led to adjuvant chemotherapy, and what share of negative results led to observation? What share led to a deferral, escalation, or documented exception?
7. Documented exception rationale. When a ctDNA-positive case did not lead to ACT, or a ctDNA-negative case did not lead to observation, was the reason recorded? This is what turns the dashboard into governance, not just reporting.
8. Action delta on chemotherapy use. What is the absolute and relative change in adjuvant chemotherapy rate compared with a matched pre-implementation cohort?
9. Action delta on oxaliplatin use. Where doublet escalation is part of the protocol, has it changed for ctDNA-positive patients?
10. Time-to-treatment where indicated. For patients receiving adjuvant chemotherapy after a positive ctDNA, has the time from surgery to first dose stayed within a clinically safe window?
11. Outcome guardrails. Recurrence-free and overall survival, monitored against the comparator, with formal safety review at predefined intervals.
12. Equity and access. Is the ctDNA pathway used uniformly across sites, payer mixes, languages, and socioeconomic groups, or only in some patient populations?
Two more metrics belong on the same page, even if they are slower to mature. Patient anxiety and decisional conflict, because risk information without decision support carries its own burden. And cost per actionable decision, not per test, because the unit economics question is about decisions changed, not assays run.
The dashboard should not ask whether ctDNA was used. It should ask whether the chemotherapy decision changed, safely.
9. The generalisable rule
The ctDNA case looks like a story about colon cancer. It is also, on reflection, a template.
Replace ctDNA with any of the following and the architecture stays intact: a multi-cancer early detection assay, an AI-based deterioration model, an FFR-CT result, an ambient documentation tool, a digital biomarker streamed from a wearable, a polygenic risk score, a companion diagnostic for a targeted therapy, a heart failure remote monitoring platform, a CAR-T eligibility test.
Each one carries a signal. Each one only earns the word care when it enters a defined decision window, with a named actor, a specific action, a clinically meaningful time window, and a measurable consequence. Each one can fail at a different rung of the ladder. Each one has a payment architecture that does or does not align who funds the signal with who captures the value.
The careless review, again and again, asks whether the technology works.
The Clinical Design review asks which decision it is supposed to change, and whether the system is built to let it.
A biomarker becomes care only when it is bound to a governed decision.
Future teardowns will change the technology. The design question will stay the same.
10. Closing
The assay returns a result. The pathway returns care.
The test is not the intervention. The governed decision is.
Don’t ask whether the biomarker works. Ask whether the treatment decision changed.
Don’t pilot the test. Pilot the care-changing unit.
— Marcos
Note & Disclaimers
Context: The Clinical Decade (and this article) explores the theoretical foundations of Clinical Design, an independent teaching framework created by Marcos Gallego. It has been developed through independent research and academic activities, and is shared here as a personal contribution to the field.
Independence: Views and materials published in The Clinical Decade are personal and independent and do not represent any employer, client, or institution.
License: Licensed under Creative Commons Attribution–NonCommercial–NoDerivatives 4.0 International (CC BY-NC-ND 4.0), unless otherwise stated.
Baxter NN, Kennedy EB, Bergsland E, et al. Adjuvant Therapy for Stage II Colon Cancer: ASCO Guideline Update. Journal of Clinical Oncology, 2022;40(8):892–910. https://ascopubs.org/doi/10.1200/JCO.21.02538
Tie J, Cohen JD, Lahouel K, et al. Circulating Tumor DNA Analysis Guiding Adjuvant Therapy in Stage II Colon Cancer. New England Journal of Medicine, 2022;386(24):2261–2272. https://www.nejm.org/doi/full/10.1056/NEJMoa2200075
Tie J, Wang Y, Tomasetti C, et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Science Translational Medicine, 2016;8(346):346ra92. https://www.science.org/doi/10.1126/scitranslmed.aaf6219
Tie J, Cohen JD, Lahouel K, et al. Circulating Tumor DNA Analysis Guiding Adjuvant Therapy in Stage II Colon Cancer. New England Journal of Medicine, 2022;386(24):2261–2272. https://www.nejm.org/doi/full/10.1056/NEJMoa2200075
Tie J, Cohen JD, Wang Y, et al. Circulating tumor DNA analysis guiding adjuvant therapy in stage II colon cancer: 5-year outcomes of the randomized DYNAMIC trial. Nature Medicine, 2025;31(5):1509–1518. DOI: 10.1038/s41591-025-03579-w. https://pubmed.ncbi.nlm.nih.gov/40055522/
Nakamura Y, Watanabe J, Akazawa N, et al. ctDNA-based molecular residual disease and survival in resectable colorectal cancer. Nature Medicine, 2024 (CIRCULATE-Japan / GALAXY 2,240-patient analysis). https://www.nature.com/articles/s41591-024-03254-6
NCCN Updates ctDNA Stance in Colon, Rectal, and MCC Guidelines. OncLive, 2025, summarising current NCCN positioning that ctDNA is recognised as prognostic but not predictive, with insufficient evidence to recommend routine ctDNA-based de-escalation or treatment decision-making outside clinical trials in the relevant adjuvant settings. https://www.onclive.com/view/nccn-updates-ctdna-stance-in-colon-rectal-and-mcc-guidelines
Tie J, Wang Y, Loree JM, et al.; AGITG DYNAMIC-III Study Group. Circulating tumor DNA-guided adjuvant therapy in locally advanced colon cancer: the randomized phase 2/3 DYNAMIC-III trial. Nature Medicine, 2025;31:4291–4300. DOI: 10.1038/s41591-025-04030-w. https://pubmed.ncbi.nlm.nih.gov/41115959/
Morris VK, Yothers G, Kopetz S, et al. NRG-GI005 (COBRA): Phase II/III study of circulating tumor DNA as a predictive biomarker of adjuvant chemotherapy benefit in resected stage IIA colon cancer. Journal of Clinical Oncology, 2024;42(3 suppl):5. ASCO GI Symposium abstract; conference reporting. https://ascopubs.org/doi/10.1200/JCO.2024.42.3_suppl.5
To YH, Gibbs P, Tie J, Loree J, Glyn T, Degeling K. Circulating Tumour DNA Guided Adjuvant Chemotherapy Decision Making in Stage II Colon Cancer—A Clinical Vignette Study. Cancers, 2023;15(21):5227. DOI: 10.3390/cancers15215227. https://www.mdpi.com/2072-6694/15/21/5227
Tie J, Wang Y, Tomasetti C, et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Science Translational Medicine, 2016;8(346):346ra92. https://www.science.org/doi/10.1126/scitranslmed.aaf6219
Nakamura Y, Watanabe J, Akazawa N, et al. ctDNA-based molecular residual disease and survival in resectable colorectal cancer. Nature Medicine, 2024 (CIRCULATE-Japan / GALAXY 2,240-patient analysis). https://www.nature.com/articles/s41591-024-03254-6
Li Y, Heer AK, Sloane HS, Edelstein DL, Tie J, Gibbs P, Barzi A. Budget Impact Analysis of Circulating Tumor DNA Testing for Colon Cancer in Commercial Health and Medicare Advantage Plans. JAMA Health Forum, 2024;5(5):e241270. https://jamanetwork.com/journals/jama-health-forum/fullarticle/2819327
Kramer A, Greuter MJE, Schraa SJ, Vink GR, Phallen J, Velculescu VE, Meijer GA, van den Broek D, Koopman M, Roodhart JML, Fijneman RJA, Retèl VP, Coupé VMH. Early evaluation of the effectiveness and cost-effectiveness of ctDNA-guided selection for adjuvant chemotherapy in stage II colon cancer. Therapeutic Advances in Medical Oncology, 2024;16. DOI: 10.1177/17588359241266164.




